Many nurses do not register their clinical trials. Our yearly report aims to shock them into action.
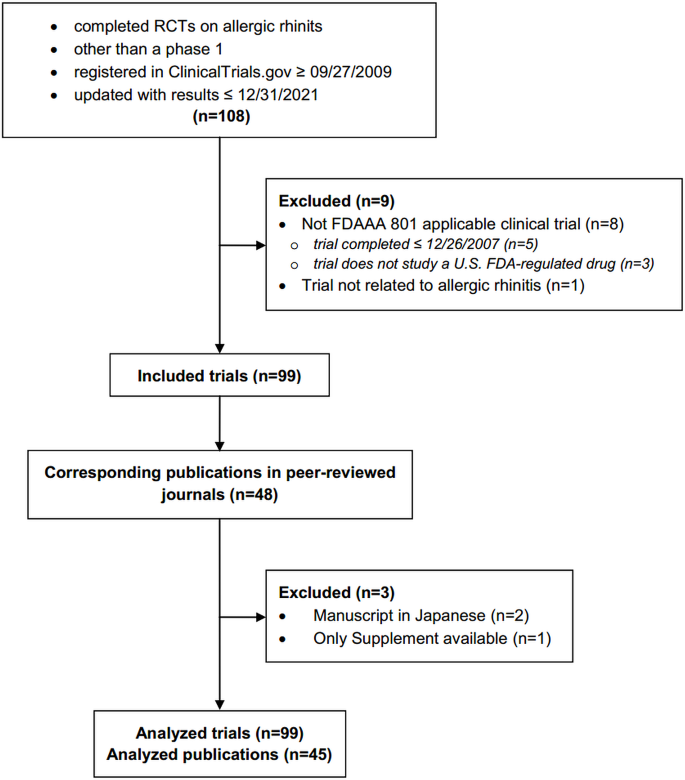
Reporting of the safety from allergic rhinitis trials registered on ClinicalTrials.gov and in publications: An observational study | BMC Medical Research Methodology | Full Text
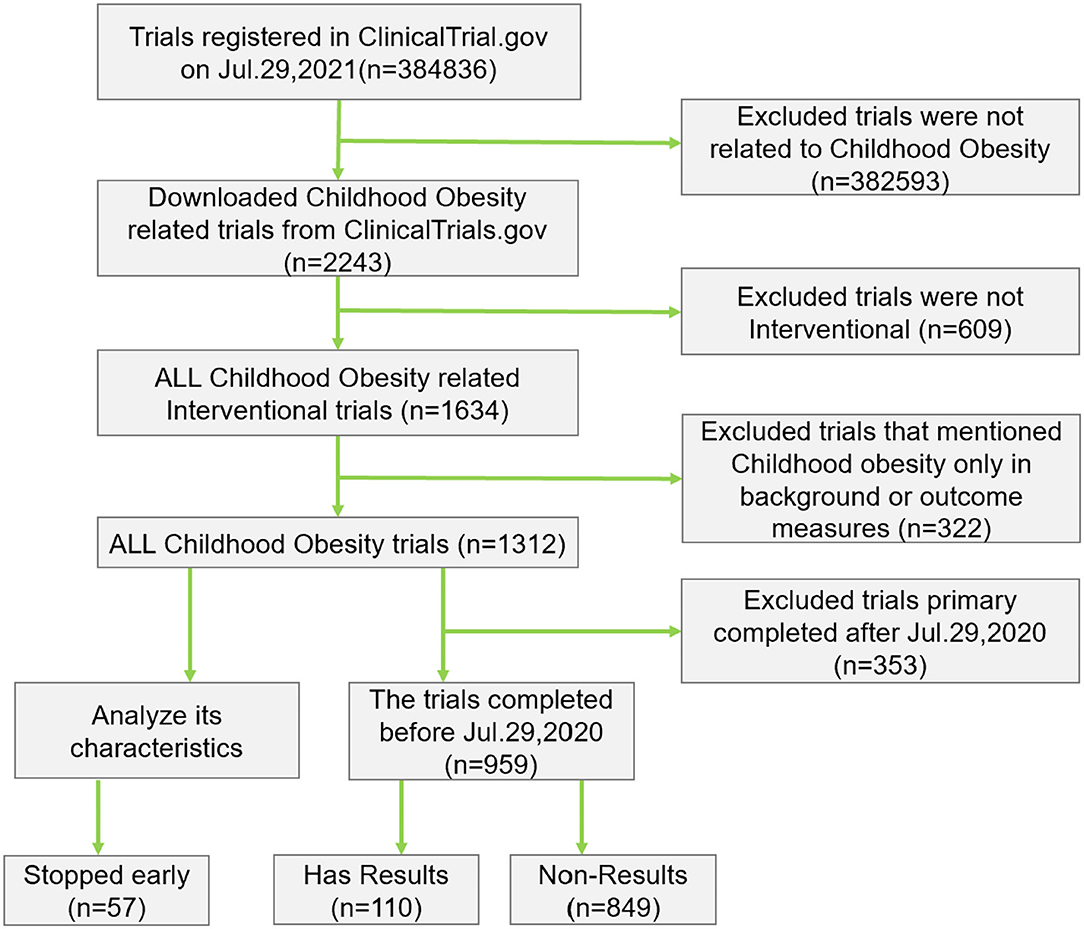
Frontiers | Results Reporting and Early Termination of Childhood Obesity Trials Registered on ClinicalTrials.gov

PDF) Using the Cochrane Central Register of Controlled Trials to identify clinical trial registration is insufficient: A cross-sectional study
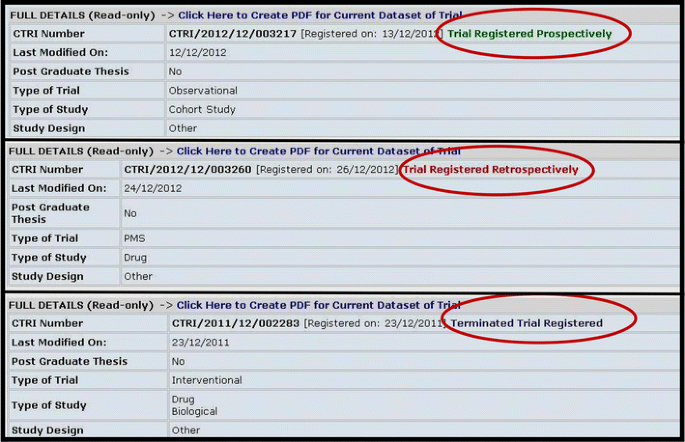
Challenges in Administering a Clinical Trials Registry: Lessons from the Clinical Trials Registry-India | SpringerLink

Regulatory Approval – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India

Registration of Clinical Trials: Background and Implementation Ida Sim, MD, PhD Project Coordinator Department of Research Policy and Cooperation World. - ppt download

Shelbourne FC Academy on Twitter: "**IMPORTANT TRIALS INFO** Please see the above information about our upcoming trials. We are NOT ACCEPTING WALK ON'S on the day. ONLY REGISTERED PLAYERS who have received

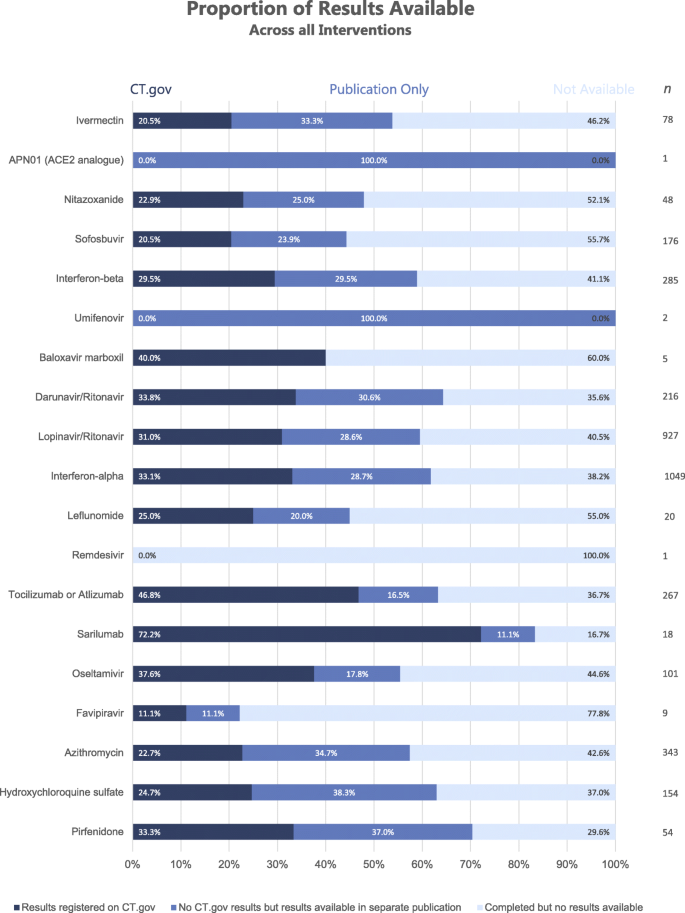
Number of registered trials by mapped drug name. Only the top 30 drugs... | Download Scientific Diagram